Binary Batch Distillation
Batch distillation is widely used in chemical processing industries when high value added, low volume chemicals must be separated. This article shows how to model and solve a multi-stage binary batch distillation problem in excel.
Example
The 50 mol feed comprising 70% mols of A and 30% mols of B is to be distilled in the multi-stage batch distillation with 5 equilibrium stages on top of the reboiler (still pot). Reflux is returned to the column as a saturated liquid with the constant reflux ratio of 1.5. Relative volatility of A is 1.8. It is desired to reduce the mols of A in still to 10%. Determine average distillate composition, final amount of liquid in still pot and total amount of distillate collected.
A material balance is done across the column and following relationship commonly known as Rayleigh Equation is derived.
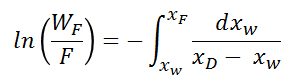
Where F (Initial mols fed in still), WF (Mols left in still at any time), xF (Initial mol fraction in feed), xW (Mol fraction in still at any time) and xD (Distillate composition at any particular time).
Equilibrium curve is obtained from relative volatility.
y = αx / (1+ x (α - 1))Operating line is obtained as following.
y = Rx /(R+1) + xD /(R+1)Relationship between xD and xW is obtained by performing stage-by-stage calculations. A value of xD is selected and stages are stepped off (for a given number of equilibrium stages) to find the value of xW. It is done for all possible values of xD ranging from 0 to 1. A plot of 1/(xD - xW) vs xW is made.
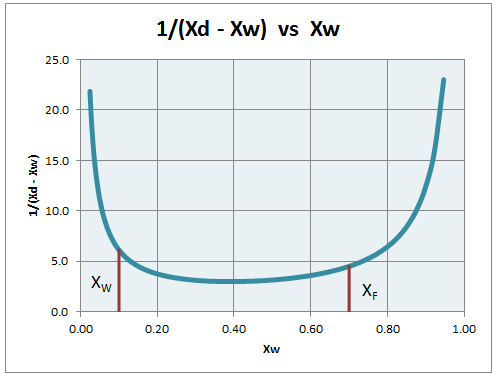
Area under curve is calculated from xW to xF by calculating area of trapezoid formed in small intervals.
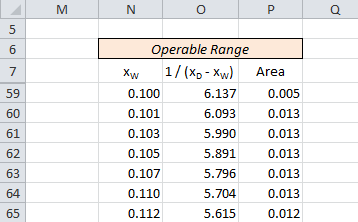
Area (Cell P59) = (N60-N59)*(O60+O59)/2)Area of all trapezoids in the range from xW to xF are added to get area under curve.
Area = 2.153Amount left in still, WF is calculated as following.
WF = F.exp(- Area under the curve)WF = 5.80 mols
Distillate mols collected is obtained as following.
D = F - WFD = 44.20 mols
Average composition of distillate collected xD is calculated as following.
xD = (xF F - xW WF)/DxD = 0.779
Resources
- Spreadsheet for Binary Batch Distillation